B2B marketing for Pharmaceutical Manufacturers built for trust and verification
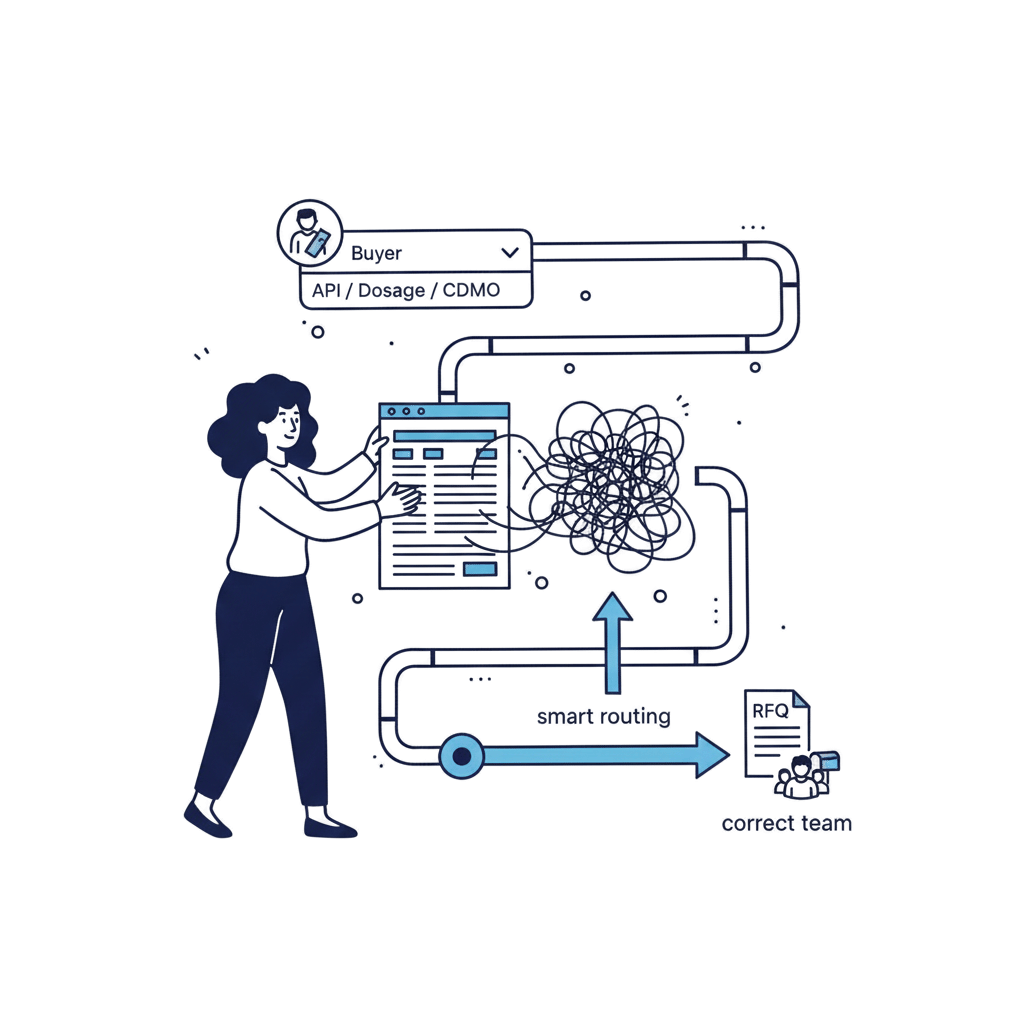
Help global buyers verify capability and send better fit RFQs
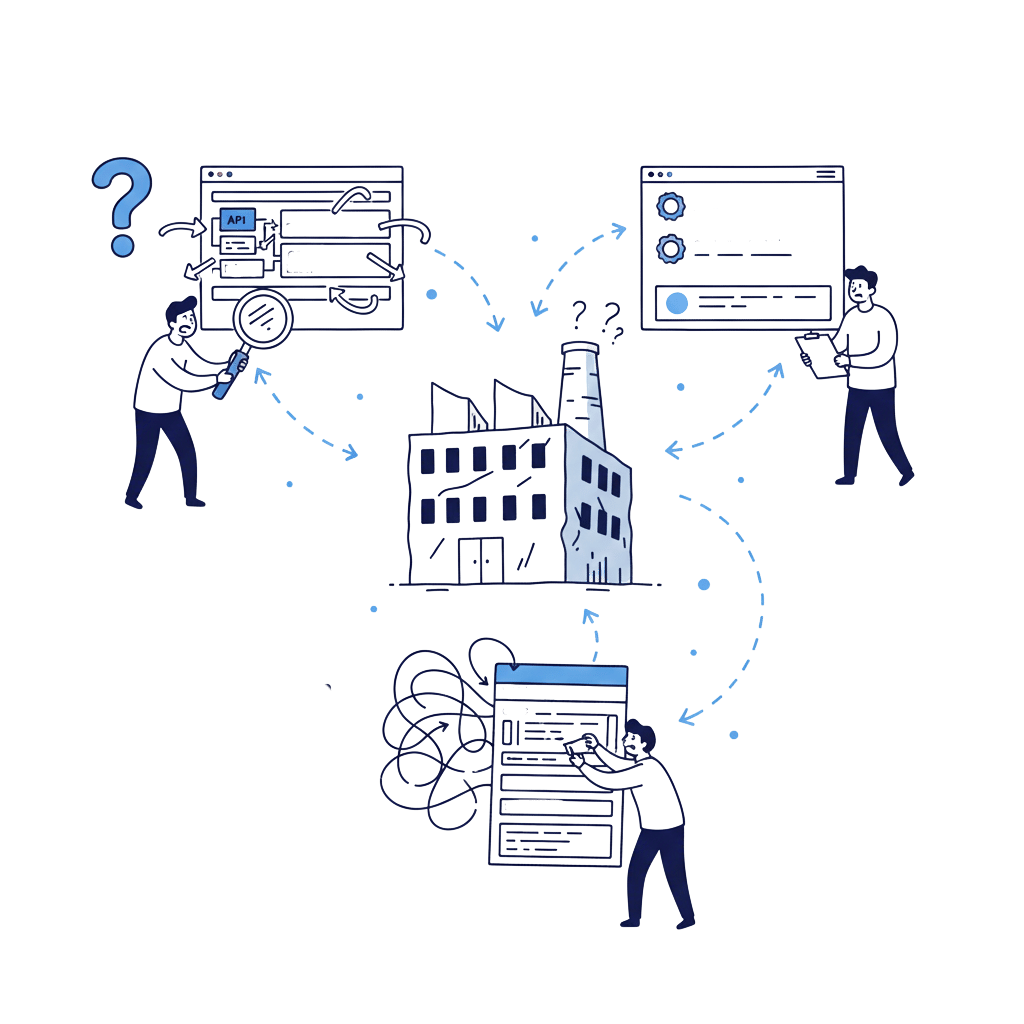
Pharma manufacturing decisions are verification led. Buyers shortlist based on capability clarity, quality approach, and how easy it is to validate and contact the right team. We deliver Healthcare Marketing for Pharmaceutical Manufacturing through credibility first websites, SEO for manufacturing intent searches, and compliance aware messaging that QA and regulatory teams can review. No exaggerated claims, no guarantees, and documentation friendly communication built for global markets.
💡 No spam. Practical next steps, even if we do not work together.